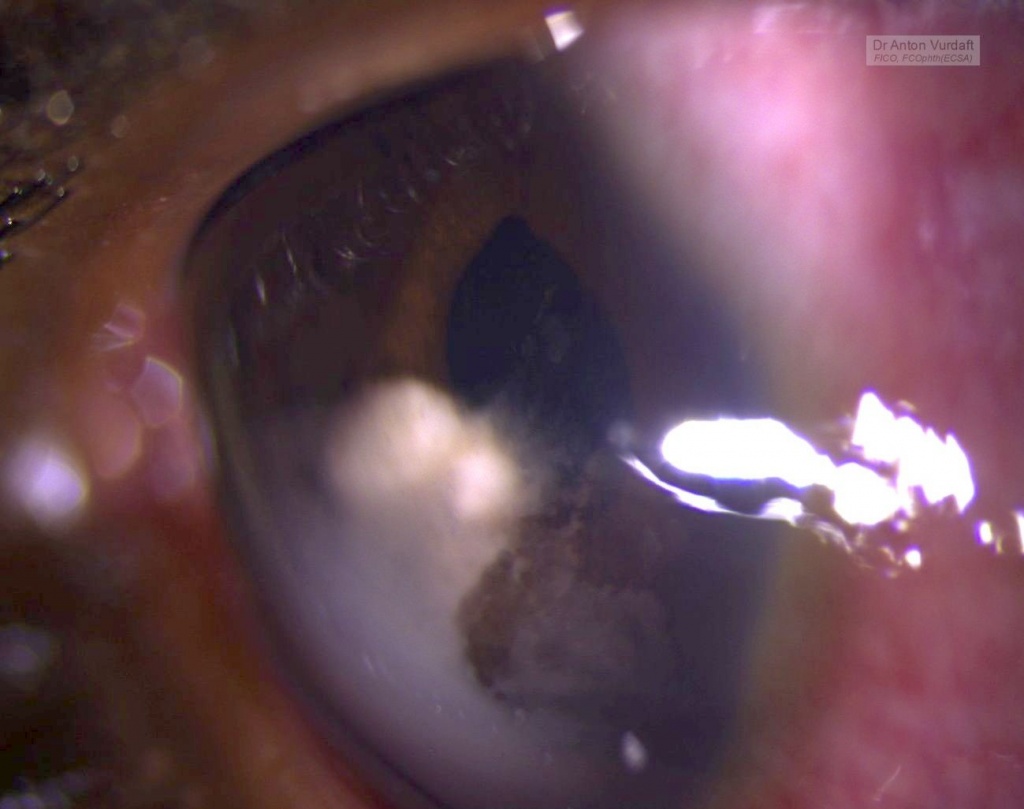
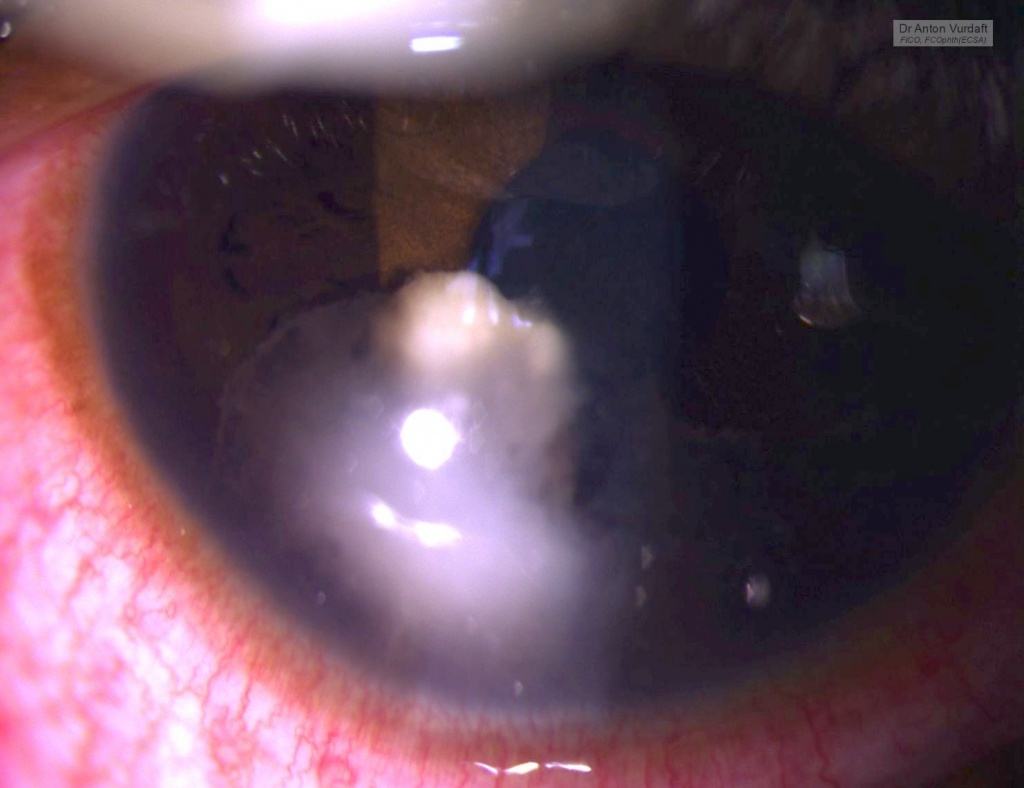
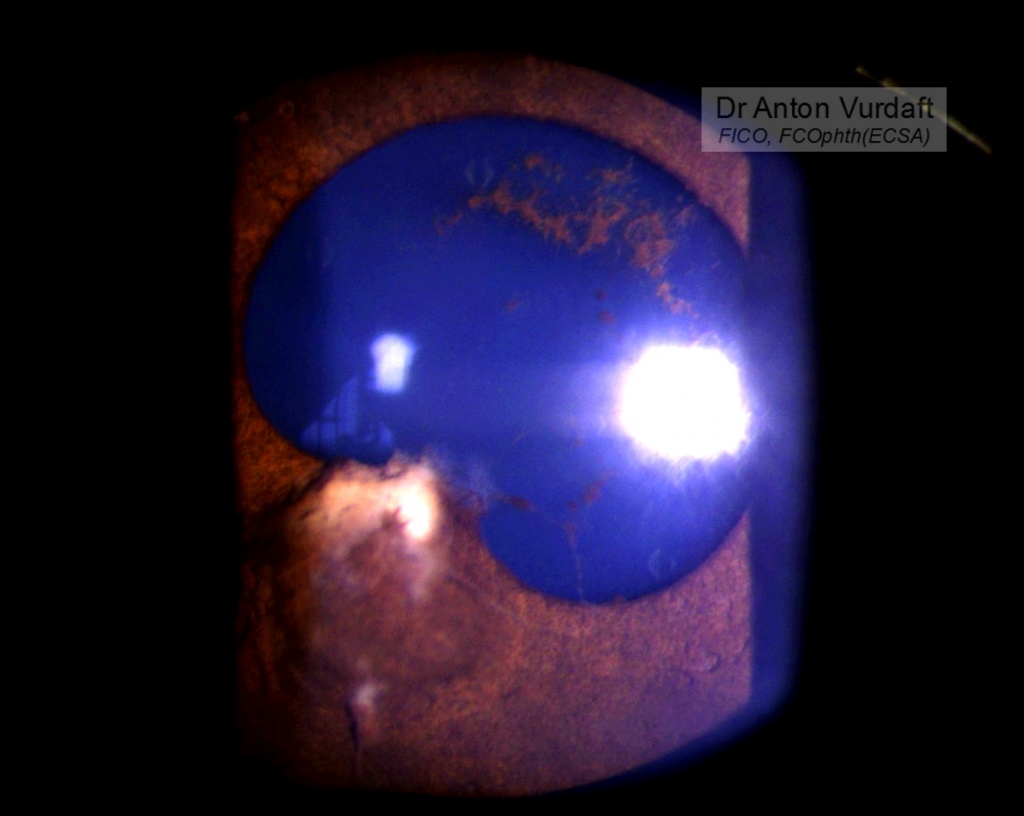
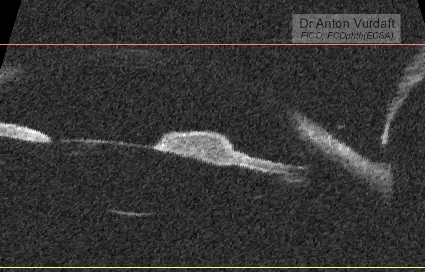
The following information is essential for those colleagues-ophthalmologists travelling abroad into the tropics, where the rate of HIV may reach up to 10-20% of a population or even more, depanding on the area. An especially for the oculoplastic colleagues. And here’s why:
The following information is essential for those colleagues-ophthalmologists travelling abroad into the tropics, where the rate of HIV may reach up to 10-20% of a population or even more, depanding on the area. An especially for the oculoplastic colleagues. And here’s why:
Rate of gloves punctures during ophthalmic procedures reaches 21.8%!
Cataract surgeons experience only 11.9% of punctures.
Oculoplastic procedures carry the biggest risk for gloves perforation: 41.67%!
Risks increase rapidly for a longer procedures.
(Glove Perforation – Miller & Apt – Arch Ophthalmol – Vol 111, Feb 1993)
When double gloved – punctures will frequently be limited by the outer pair of gloves. Two layers may also reduce the amount of blood actually got into the skin with the sharps.
So, you have had an incident. You’ve pricked yourself with a suture needle or any other sharp during the surgery. What are your steps?
1) Stop the surgery immediately.
2) Wash your hands with soap. The CDC guidelines wouldn’t advise spirit or povidon iodine wiping, but the temptation is highest.
3) Scrub in and complete the case.
4) This is definitely the end of your operating day today, because:
5) You need to get the status of HIV of the patient as fast as possible. Ideally you have got the test done for all your patients before the surgeries.
6) The patient found positive (by fresh test or via previous history).
7) You test yourself as well (as per protocol, the post-exposure prophylaxis is not indicated if the one is already HIV-positive).
8) You are negative. Check your liver function tests and creatinine.
9) Initiate the postexposure HIV prophilactic therapy (“PEP”). This is a set of 3-4 HAART drugs. Please, take the best PEP combination with yourself to your trips, regardless of the stocks available on sites (you never know their quality!).
10) This is for 28 days, and not without side effects, such as transitory jaundice, diarrheoa/vomitting (depending on therapy applied) and rash/pruritus.
10) Keep monitoring your side effects with the HIV-specialist. Monitor liver function tests PRN.
What works for you in this situation:
1) The risk for HIV transmission is very low by default – about 0,3% (that is good)
2) You used two pairs of gloves (that is clever in these regions!)
3) Needles from sutures carry less risk for transmission than hollow needles for injections
4) The patient might be on HAART already, this reduces his viral load to less than 1700 copies per ml (and that is awesome, risks go even lower)
5) The PEP, which you have started within two hours (24-48 hours at most!, the more – the worse), reduces the remaining lowest chances to close to zero.
What works against you:
1) High HIV viral load in the source patient (more than 1700 copies per ml increase chances for transmission 16 times, according to some papers).
2) Late initiation of PEP (studies on monkies show, that late start – after 48 hours – is almost useless, as it allows more than 50% “lucky” guys be infected still).
P.S. I am not talking about the situation, when the patient is found negative, though it can be a very tricky question. Remember, there are window periods for the HIV to become detectable by routine express tests.
If there is even a slight level of uncertainty – you must start the PEP.
Stay safe!